Health
Electronic noses for early diagnoses of diseases by smell

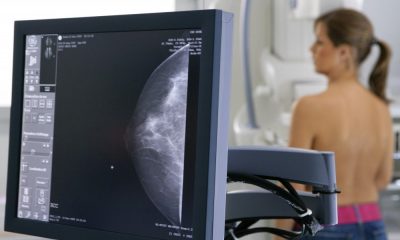
We may soon be able to obtain easy and early diagnoses of diseases by smell. This week researchers found one odour-sniffing machine was as good as a mammogram at detecting breast cancer – and many other devices capable of spotting other diseases may be on the way.
“I may sound crazy but I’m not,” writes Joanie, on an online support forum for people affected by cancer. She relates how, while her husband suffered from prostate cancer, she could smell “an odour similar to decay”. It went away with the cancer, but in 2012 she was alarmed to smell the putrid stench once again. Not long afterwards, Joanie herself was diagnosed with lung cancer.
Although many cancer sufferers and their relatives do not notice a nasty smell, Joanie’s experience is not unusual. “I’ve had numerous people writing to me about this,” says George Preti from the Monell Chemical Senses Center in Philadelphia. “I’ve had lots of notes from nurses and researchers in the area, but they’re mostly anecdotal reports.”
Throughout history, doctors have sniffed their patients’ breath, urine, stool and other bodily fluids to help with diagnoses. A 2011 review article featured “smelling notes” of dozens of diseases. Yellow fever is said to smell like a butcher’s shop, liver failure like raw fish, and typhoid like freshly baked brown bread.
A recent experiment also provides some evidence that people give off a bad smell as they fall sick.
A team led by Mats Olsson at the Karolinska Institute in Stockholm dressed eight healthy volunteers in tight cotton T-shirts and injected one half of the group with a placebo and the other half with a chemical that gave them a mild flu-like reaction. A month later, the participants came back and were injected with the other solution – the one they had not received the first time. All the T-shirts were then collected and patches from the underarm area were cut out and placed in squeeze bottles. These were used to supply puffs of air to a panel of smelling volunteers. “They rated the T-shirts with regards to intensity, unpleasantness and healthiness,” says Olsson. “And for all these measures it turned out that the so-called sick group smelled more aversively.”
Although Olsson admits that the study – published in the journal Psychological Science – was very small, he believes ill people may emit a chemical “cue” to warn others of contagion, or possibly to signal they need help. Bad smells exist for a reason, agrees Val Curtis from the London School of Hygiene & Tropical Medicine. “The disgust system in the brain has evolved to make us stay away from things that might make us sick. The most dangerous place on the planet is other people.”
For Mats Olsson, “the new thing and interesting thing” about his T-shirt study is that human beings seem capable of sniffing out disease early on – long before we start smelling like a butcher’s shop or raw fish.
For cancer, early detection is crucial, but the symptoms that prompt a visit to the doctor’s surgery often don’t emerge until the disease is advanced. George Preti gives the example of ovarian cancer, which has become the focus of his own work. “If you picture a peppercorn and an onion, the difference in size between those two is the difference between when it should be diagnosed and when it’s traditionally diagnosed,” he says.
He hopes that ovarian cancer’s smell could help doctors arrest the disease at the peppercorn stage. As part of everyday metabolism, our bodies produce chemicals which evaporate into the air. Cancerous cells, Preti says, have a different metabolism from normal cells, and therefore emit a different pattern of chemicals, a different odour. While it’s too subtle for most human noses, dogs have been trained to identify multiple types of cancer, as well as diabetes. (Humans have around five million scent receptors, but dogs have up to 300 million.)
Preti is working with the Penn Vet Working Dog Center, which is training four dogs to detect ovarian cancerous tissue. He says they can do this with greater than 90% accuracy. However, the idea is not that these dogs will get jobs in the oncology departments of hospitals. Instead, the Penn Vet dogs will help Preti identify the unique “odour signature” of ovarian cancer. “They’re sort of a four-legged bio-assay,” he says.
Once the odour signature or profile of ovarian cancer is determined, it will be used by Preti’s colleague Charlie Johnson to program sensors in a device sometimes called an “electronic nose” or “e-nose”. Johnson’s prototype mimics the receptors in our nose with hundreds of carbon nanotubes, each with a strand of DNA attached. These strands are able to transpose the mix of chemicals in the air into an electronic signal. “You can have the power of a dog’s nose on this small chip the size of your fingernail,” Preti says. It’s this device, rather than the dogs, that might end up on hospital wards.
But dogs aren’t the only animals that can smell cancer. So, apparently, can fruit smiles. A recent paper in Nature showed that the scent of cancer samples prompted a particular pattern of activity on the flies’ antennae. The paper’s author, Martin Strauch at the University of Konstanz in Germany, is planning to identify which receptors on the antennae respond to the cancerous smells. Then he hopes to grow his own antennae in a petri dish, for use in an e-nose. “The biological sensors have been optimised by evolution – they are quite good,” he says.
Electronic noses of other kinds are already being put to work in a range of different fields, from checking for poison gas in war zones to ensuring the quality of beer. They are now on the verge of being set medical tasks, following tests on a device called BreathLink, which is designed to check for breast cancer.
The device, developed by a company called Menssana, starts by building a profile of the ambient gas in a room. A patient in the room then breathes into a tube for two minutes. The composition of her breath is laid over the ambient room profile, and whatever’s left over is compared to the known profile for breast cancer held in a database in the US. The results of the latest tests on BreathLink show it has similar diagnostic power to a mammogram, but women don’t have to undress and are not exposed to X-rays.
The results also come through within 10 minutes, instead of two weeks. Dr Roy Lalisang, who tested the machine at the University of Maastricht Medical Centre in Holland, says further tests are required, but it could be ready for service in the next couple of years. BreathLink could also, potentially, be used to test for tuberculosis.
Another company, Owlstone, also hopes to bring a device to market in roughly the same time-frame, which would be used to screen patients for bowel cancer and other digestive diseases. Called Lonestar, this device would prevent some patients having to undergo an uncomfortable colonoscopy.
As well as screening patients, medical sniffing devices can potentially be used to monitor them patients, as well as screening them. Krishna Persaud at the University of Manchester has been testing one that monitors victims of severe burns for signs of infection – if his e-nose sniffs an unwelcome odour, doctors can react immediately, instead of waiting three days for the sample to be analysed in a lab.
Along-term goal for the sector is to get easy-to-use diagnostic tools into the home… and into our pockets. With the ever-growing sophistication of mobile phones – some of the latest models have temperature and humidity sensors – the idea of routinely breathing on to your phone to check for disease is not science fiction. The chip already exists, and getting it into a phone is straightforward engineering or electronics, says Billy Boyle of Owlstone, whichwas a recent runner-up ina competition sponsored by Nokia.
For lots of patients these new tools cannot arrive in hospitals and doctors’ surgeries too quickly.
Roy Lalisang says that since the results of the BreathLink study were published last Wednesday his phone has not stopped ringing – most of the callers being women who are sick of getting prodded and pummelled in mammograms.
“In my daily practice, I so frequently hear from women who have to undergo mammography,” he says. “They say, ‘Oh, when is the next one? In one year? Is it really necessary because it’s so painful!’ I always say: ‘If men had to undergo this examination they would have found another test many years ago.’
“Women will not undergo mammography if this is a comparable screening test.”
Source: BBC
You may like

Gal Gadot Gives Birth to Third Child

Ariana Grande Is Giving Away One Million Dollars for a Good Cause

Potato Nests with Peas, Ham and Cream Cheese

So, you can get yeast infection from biting your nails